Document Type : Original Research Article
Authors
1 Department of Chemistry, College of Science, University of Thi-Qar, 64001 Nassiria, Iraq
2 Department of Pathological, College of Science, University of Sumer, Iraq
Abstract
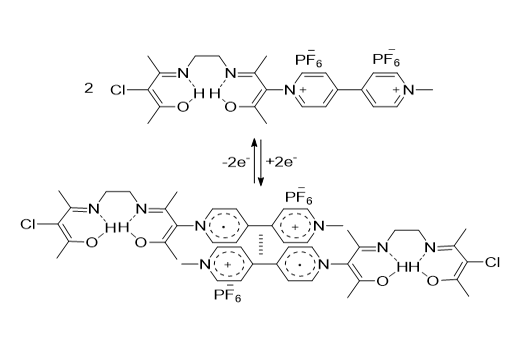
In this work, the viologen derivative was prepared by reacted of bis (3-chloroacetylacetone) ethylenediimine (AN-Cl) with mono-methylviologen (C1V+). The switching motion between dicationic viologen compound C1V2+AN-Cl.2PF6- and diamagnetic intermolecular π-dimer (C1V+AN-Cl. PF6-)2 was triggered through the reduction by 1 electron/viologen unit. This molecular switch was followed by UV-Visible absorption spectrometry. The viologen compound C1V2+AN-Cl.2PF6- was titrated with Zn2+, Cu2+, Co2+, and Fe2+ ions to afford metal complexes in the solution of M2+-C1V2+AN-Cl.2PF6-. Moreover, four molecular switches were attained from those metal complexes (M2+-C1V+AN-Cl. PF6-)2 by the chemical reduction stimuli including the characterization of complexes of M2+-C1V12+AN-Cl.2PF6- were performed by UV-Visible absorption spectroscopy. It was noticed that ratio of metal to the ligand in the complexes of Zn2+, Cu2+, Co2+, and Fe2+ was 1:1. Switching properties of the novel viologen derivatives with intermolecular dimer were studied by the reduction with activated zinc powder and followed by the absorption spectroscopy. After the 1e- reduction viologen unit in C1V12+AN-Cl.2PF6- and M2+-C1V12+AN-Cl.2PF6-, intermolecular dimerized viologen radicals occurred.
Graphical Abstract
Keywords
- Bis (3-chloroacetylacetone) ethylenediimine
- 4,4'-bipyridine
- Viologen
- Intermolecular π-dimerized C1V2+AN-Cl.2PF6-
Main Subjects
Introduction
Viologens are organic salts of bipyridines quaternized at one or two nitrogen atoms. If the two substituents at two nitrogen atoms are the same, they are called 1,1'- disubstituted-4,4'-bipyridinium salts; if they are different, they are called 1-substituent-1'-substituent'-4,4'-bipyridinium salts.
When both substituents are methyl groups, the resulting dicationic molecule is known as methyl viologen. It was discovered to be an effective herbicide after being studied as a redox indicator in biological investigations. Michaelis discovered the term "viologen" in 1933 after observing the violet color that resulted from the reduction of 1,1'-dimethyl-4,4' bipyridinium salt (DMV2+) by one electron to generate a radical cation (as a dimer) [1].
Viologens had been widely used as an antibacterial agent or herbicides. They are used, for example, as an active element in electrical display devices, diodes and transistors, memory devices, and molecular machine, electrodes for supercapacitors and batteries catalysts for hydrogen production and gas storage and separation, .pigments for sensitizing solar cells.
The main characteristics of viologens that make them important and essential building blocks for many materials can be summarized as follows: reduction of viologens to colored positive free radicals, and then to neutral species that can occur sequentially and reversibly within accessible potentials. Three oxidation states of cationic state radical cation and neutral state are and thermodynamically stable.
Experimental
Synthesis of 1-methyl-4,4'-bipyridinium (C1V+) [2]
Methyl iodide (0.39 mL, 6.4 mmol) was added with stirring into a solution of 4, 4'- bipyridine (1gm, 6.4 mmol) in 10 mL of di chloromethane, and then the solution was stirred at room temperature for 24 hours. The produced precipitate was collected by filtration, washed with dichlioromethan and diethylether, recrystallized from methanol, and then dried under vacuum to afford C1V+.I- as a yellowish-orange precipitate, yield: 67%. M.p= 252 οC.
C1V+.I- , 1H-NMR (500 MHz, DMSO-d6), δ (ppm): 9.16 (d, J = 6.1 Hz, 2H, Ha), 8.86 (d, J = 4.9 Hz, 2H, Hb), 8.63 (d, J = 6.1 Hz, 2H, Hc), 8.04 (d, J = 4.9 Hz, 2H, Hd), and 4.40 (s, 3H, He).
Anion exchange
From Iodide forms to hexafluorophosphate form, the C1V+.I- was dissolved in the minimum volume of distilled water. Upon the addition of saturated aqueous KPF6 to the previous solution, the hexafluorophosphate salt of viologen derivative has been precipitated. The precipitate was collected by filtration and washed with distilled H2O to afford the PF6- form: C1V+.PF6- as a white precipitate.
Synthesis of bis (3-chloroacetylacetonato) ethylenediimine (AN-Cl)[3]
Ethylenediamine (0.6 mL, 0.5315 g) in 2 ml ethanol was added gradually to 3-chloro acetylacetone (2 mL, 2.38 g) in 2 mL ethanol placed in ice bath. The resulting mixture was stirred at room temperature for 24 h. The reaction progress was controlled by TLC (eluent = 4:1 Methanol: Benzene). The reaction mixture was left to dry under air atmosphere, and then under vacuum at room temperature.
The produced precipitate was washed with distilled water, m.p. 134 °C, yield 71.8 %.
Syntheses of final viologen derivative of C1V2+AN-Cl.2PF6-
C1V1+.I- (1 gm, 3.35 mmol) was dissolved in DMF with stirring. When the solution becomes clear, AN-Cl (0.98 gm, 3.35 mmol) was added to the solution and the reaction.
Thermal analysis of C1V2+AN-Cl.2PF6- ligand
The Thermal behavior of C1V2+AN-Cl.2PF6- thermal gravimetric analysis (TGA) was used to study the ligand, which was recorded in nitrogen gas at a constant heating rate of 15 ˚C/min. The decomposition pattern of C1V2+AN-Cl.2PF6- ligand occurs in three phases, as displayed in Figure 2 and Table 2. The following were these phases: the initial phase of C1V2+AN-Cl.2PF6- starts at 45.99 ˚C and end at 101.17 ˚C with weight loss of 4.538%. This first stage includes the loss of two water molecule. The following phase begins at 196.37 ˚C and end at 344.92 ˚C which correspond to the losses of PF6 and methyl group. The final phase begins at 345.97 ˚C and ends at 599.39 ˚C and is due to the loss of ethylenediamine and chlorine atom and this correspond to weight loss of 15.588% [19].
Determination of rate constants, half-lives, and thermodynamic parameters using TG analyses
All thermal analysis stages can be considered as first order reactions. Therefore, using the first-order reaction rate equation, activation energy and other thermodynamic parameters were determined for all phase transformations [20].
ln (1-x) = -kt (1)
Equation 1 is plotted (time is x-axis and ln(1-x) is y-axis) and each line's slope provides the rate constant (k) for a specific TG phase, (Figures 3-5), and then the half-life time (t1/2) was determined using Equation 2. Table 3 indicates values of k and t1/2.
t1/2 = 0.693/k (2)
Equation 3 illustrates how to determine kinetic parameters using a modified Coats and Redfern model [21-23]:
Ln [-ln(1-x)] = ln ART2/βEa – Ea/RT (3)
Where, β is the heating rate (10 °C/min and 20 °C/min), A is the pre-exponential factor, R is general gas constant (8.3143 Jmol-1K-1), Ea is the activation energy, and T is the temperature (K). The activation energy is calculated by plotting graphs of ln[-ln(1-x)] against 1000/T for each TG phase (Figures 6-8). Further thermodynamic parameters (∆H, ∆S, and ∆G) are calculated using basic thermodynamic equations [24,25]. Values obtained for each phase (Table 3) proved that all TG phases are non-spontaneous (positive ∆G values) endothermic (positive ∆H values) reactions.
The reduction of C1V2+AN-Cl.2PF6- ligand
For the first time, C1V2+AN-Cl.2PF6- was interestingly reduced by solvation only in DMF to afford the intermolecular dimerized viologen radicals of C1V●+AN-Cl.2PF6- [26,27]. Also, the reduction was performed by activated zinc powder. Figure 9 illustrates the reduced C1V2+AN-Cl.2PF6- by DMF and activated zinc powder.
At dissolving C1V2+AN-Cl.2PF6- in DMF, purple solution is obtained. This novel result refers that C1V12+AN-Cl.2PF6- molecules have been reduced by only solvation to viologen C1V12+AN-Cl.2PF6- that quickly and spontaneously π-dimerized to (C1V+AN-Cl.2PF6- )2. The absorption bands occurred at 358 nm and 424 nm at dissolving in DMF are assigned to dimerized and non-dimerized viologen radicals of C1V2+AN-Cl.2PF6- (Figure 9, black spectrum and Table 4).
At reduction by activated zinc powder, deeper purple solution has been obtained compared with solution in DMF only. This indicates to more reduction happened by activated zinc powder which is meaning more formation of intermolecular π-dimer among radical viologens.
Therefore, all absorptions after reduction by activated zinc powder have notably increased. The absorption peak at 424 nm is attributed to viologen radicals C1V●+AN-Cl.2PF6- and those noted at 358 nm and 860 nm are absolutely related to formation of intermolecular π-dimer of viologen radicals, i.e. formation of (C1V+AN-Cl.2PF6-)2 species (Figure 9 (red line), Table 4, and Scheme 1).
Reduction of Zn2+-, Cu2+-, Co2+, and Fe2+- C1V2+AN-Cl.2PF6-complexes
The transition metals complexes of C1V2+AN-Cl.2PF6- have been formed after addition 1 equivalent of tetrafluoroborate salts of Fe2+, Co2+, Cu2+, and Zn2+ in DMF. The viologen units within these complexes are also reduced by solvation in DMF and activated zinc powder. The spectra of these reduced compounds' UV-Visible absorption solutions are shown in Figures 10-13. Their data are listed in Tables 5-8, respectively.
The solutions of metal complexes with C1V2+AN-Cl.2PF6- in DMF are also purple. This result indicates that the formation of complexes of C1V2+AN-Cl.2PF6- with metal complexes does not prevent the interaction among viologen radicals to affoard the intermolecular dimerization among two viologen radicals belonging to two metal complexes. The purple solutions of Fe2+, Co2+, Cu2+, and Zn2+ complexes showed, respectively, the absorption bands 358 nm and 862 nm, 358 nm and 862 nm, 358 nm and 858 nm, and 358 nm and 863 nm. These absorption peaks are assigned to the intermolecular π-dimerization among each two viologen radicals of two adjacent complexes [26-30]. The absorption peaks occurred at 423 nm, 405 nm, 405 nm, and 401 nm in absorption spectra of Fe2+, Co2+, Cu2+, and Zn2+ complexes are attributed to non-dimerized viologen radicals [31-35]. The reduction of metal complexes by activated zinc powder resulted deeper purple solutions, i.e. increased absorbances at mentioned the maximum wavelengths. This is related to formation more viologen radicals, and then more π-dimers of these radicals, see Figures 10-14, Tables 5-8, and Scheme 2.
UV-Visible absorption spectroscopy titration of C1V2+AN-Cl.2PF6- with Zn2+, Cu2+ and Fe2+ ions
The novel viologen compound C1V2+AN-Cl.2PF6- was titrated using zinc tetrafluoroborate salts (II), copper (II), and iron (II) in ACN and followed by UV-Visible absorption spectroscopy, (Figures 3.20-3.22), respectively.
The spectrum of absorption of 0.07 mM C1V2+AN-Cl.2PF6- was initially recorded. Secondly, the absorption spectrum was recorded after each addition of Zn2+, Cu2+, and Fe2+ ions to viologen compound solution. These spectra are demonstrated in Figures 3.14 (A), 3.15 (A), and 3.16 (A) respectively. Huge concentration of the titrant Zn2+, Cu2+ and Fe2+ solution (50 mM) was used to avoid the effect of dilution during titration. The recorded absorptions of this solution was plotted versus added amount of Zn2+, Cu2+, and Fe2+ certain wavelengths and presented in Figures 14 (B and C), 15 (B and C), and 16 (B, C and D), respectively.
The absorption spectra of C1V2+AN-Cl.2PF6- with different equivalents of Zn2+ are presented in Figure 2.14 (A). The spectrum of 0.07 mM C1V12+AN-Cl.2PF6- showed three absorption peaks in ACN which are noted at 259 nm, 311 nm, and 394 nm. These peaks could be attributed to ligand C1V2+AN-Cl.2PF6- centered (LC, π–π*, and n–π*) [36] absorptions. Upon titration of C1V2+AN-Cl.2PF6- with Zn2+ (formation of the complex: Zn- C1V12+AN-Cl.2PF6-), the intensities of these peaks increased gradually with the increase of the added amounts of the metal ion. This increase in peak intensities continues till the ratio of metal: ligand is 1:1, and then constant intensities are noted for several later equivalents. Therefore, increased amounts of the complex have been formed before this ratio till getting the higher form of the complex at this ratio (Figures 14 (B and C) and Scheme 3) [37].
The 0.07 mM C1V12+AN-Cl.2PF6-solution was titrated with 50 mM of Cu(BF4)2.6H2O in ACN and followed by absorption spectroscopy, as depicted in Figures 15 (A). The recorded absorptions at 258 nm and 312 nm of this solution against different amounts of added metal ions are plotted and presented in Figures 15(B and C). Similar notes and results appeared like those in the case of titration with Zn2+ ion.
The 0.07 mM C1V12+AN-Cl.2PF6-solution was titrated with 50 mM of Fe(BF4)2.6H2O in ACN and followed by absorption spectroscopy, as shown in Figure 16 (A). The recorded absorptions at 275 nm, 311 nm, and 385 nm of this solution against different amount of added metal ion are plotted and presented in Figures 16 (B, C, and D). Similar notes and results appeared like those in case of titration with Zn2+ ion.
Conclusion
In solution, Zn2+, Cu2+, and Fe2+ complexes with C1V2+AN-Cl.2PF6- are prepared. The Zn2+, Cu2+, and Fe2+ complexes with C1V2+AN-Cl.2PF6- are characterized by UV-Visible absorption spectroscopy. The metal ligand ratio is 1:1. We also achieved the main target of this thesis which is the design of one novel molecular switch. Switching from dicationic viologen C1V2+AN-Cl.2PF6- and M2+-C1V2+AN-Cl.2PF6- to intermolecular dimerized viologen (C1V+AN-Cl.2PF6-)2 and intermolecular dimerized complexes (M2+-C1V+AN-Cl.2PF6-)2.
Acknowledgements
The financial support for this research was provided by Ministry of Higher Education Malaysia under FRGS Grant no. 2019-0147-103-02.
Conflict of Interest
The authors declare that there is no conflict of interest.
Orcid:
Jassem Israa: https://orcid.org/0000-0002-9609-0498
Abdul-Hassan Wathiq: https://orcid.org/0000-0003-1297-3822
Flafel Ibrahim: https://orcid.org/0000-0002-9407-4996
----------------------------------------------------------------------------
How to cite this article: Israa. A. Jassem*, Wathiq S. Abdul-Hassan, Ibrahim A. Flafel. Novel molecular switches based on viologen ligand and its transition metal complexes. Eurasian Chemical Communications, 2023 5(9), 758-775. Link: https://www.echemcom.com/article_171848.html
----------------------------------------------------------------------------
Copyright © 2023 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
_(1).png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
_(1).png)
.png)
.png)
_(1).png)
.png)
.png)
.png)
.png)
